A) [Pt(NH3) 4]2+
B) [Ni(NH3) 4]2+
C) [Pt(NH3) 2Cl2]
D) [Pt(NH3) Cl3]-
E) [Ni(NH3) 3Cl]+
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electron configuration of an Fe2+ ion is
A) [Ar]4s24d4.
B) [Ar]4s23d6.
C) [Ar]3d3.
D) [Ar]3d5.
E) [Ar]3d6.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The best name for the complex shown below is 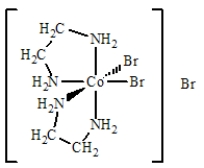
A) cobalt(III) bis(ethylenediamine) bromide.
B) dibromobis(ethylenediamine) cobalt(III) bromide.
C) dibromidedi(ethylenediamine) cobalt(III) bromide.
D) dibromodiethylenediaaminecobalt(III) bromide.
E) tribromobis(ethylenediamine) cobalt(III) .
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A complex with the composition [MA2B2]X2 is found to have no geometrical isomers.Both A and B are monodentate ligands.The structure of the complex is
A) linear.
B) square planar.
C) tetrahedral.
D) octahedral.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of these complex ions would absorb light with the shortest wavelength?
A) [Co(H2O) 6]2+
B) [Co(NH3) 6]2+
C) [CoF6]4-
D) [Co(CN) 6]4-
E) [Co(en) 6]2+
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the coordination compound [Cr(NH3) (en) 2Cl]Br2, the coordination number (C.N.) and oxidation number (O.N.) of the metal atom are, respectively,
A) C.N.= 6; O.N.= +4.
B) C.N.= 6; O.N.= +3.
C) C.N.= 5; O.N.= +2.
D) C.N.= 4; O.N.= +2.
E) C.N.= 4; O.N.= +3.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Two complex ions containing Ni are [Ni(NH3) 6]2+, which is blue, and [Ni(en) 3]2+, which is purple.Which one of these statements is true?
A) The crystal field splitting energy ( ) is greater for [Ni(NH3) 6]2+ than for [Ni(en) 3]2+.
B) [Ni(en) 3]2+ absorbs light in the violet region of the spectrum.
C) Both complex ions are diamagnetic.
D) The energy of the photon absorbed by [Ni(en) 3]2+ is greater than that absorbed by [Ni(NH3) 6]2+.
E) The wavelength of the light absorbed by [Ni(en) 3]2+ is greater than the wavelength absorbed by [Ni(NH3) 6]2+.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following complexes has optical (chiral) isomers?
A) [Co(H2O) 4Br2]+
B) [Co(en) Cl4] -
C) [Co(en) (H2O) 4] 3+
D) [Co(en) 2Cl2]+
E) Co(H2O) 3Cl3
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Labile complexes are coordination complexes that
A) are labeled with a radioactive isotope.
B) are thermodynamically stable.
C) are thermodynamically unstable.
D) undergo rapid ligand exchange reactions.
E) undergo very slow ligand exchange reactions.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the coordination compound [Co(en) 2Cl2]Cl, the coordination number and oxidation number of the central atom are, respectively,
A) 4, +3.
B) 6, +2.
C) 4, +2.
D) 6, +3.
E) 4, +1.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
A molecule or atom that accepts an electron pair to form a coordinate covalent bond is called a Lewis _______.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In K4[Fe(CN) 6], how many 3d electrons does the iron atom have?
A) 3
B) 4
C) 5
D) 6
E) 7
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the oxidation number of Co in the complex [Co(H2O)4Cl2]Cl?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The total number of electrons in the 3d orbitals of Co3+ is
A) 4
B) 5
C) 6
D) 7
E) 10
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following choices can function as a bidentate ligand?
A) PH3
B) Cl-
C) CN-
D) (-OCH2CH2CH2O-)
E) CO
G) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the coordination number of cobalt in [Co(NH3)6]Cl3?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ethylenediaminetetraacetic acid (EDTA) is
A) not useful as a chelating agent.
B) an effective antidote for heavy metal poisoning (e.g., Pb2+ and Hg2+) .
C) a monodentate ligand.
D) known to form unstable complex ions with Fe3+, Hg2+, and Zn2+.
E) known to form complexes with platinum that inhibit the growth of cancerous cells.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How would you expect the molecule 1,10-phenanthroline (shown below) to function as a ligand? 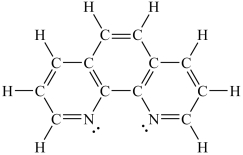
A) It would be expected to be a monodentate ligand.
B) It would be expected to be a bidentate ligand.
C) It would be expected to be a tridentate ligand.
D) It would be expected to be a tetradentate ligand.
E) It would not be expected to function as a ligand.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The correct formula for pentaamminechloroplatinum(IV) chloride is:
A) [Pt(NH3) 5Cl]Cl3
B) [Pt(NH3) 5Cl]Cl2
C) [Pt(NH3) 5]Cl3
D) [Pt(NH3) 5Cl]Cl4
E) [Pt(NH3) 5]Cl4
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the coordination number (C.N.) and oxidation number (O.N.) of the metal atom in the coordination compound [Cr(NH3) 2(en) Cl2].
A) C.N.= 4; O.N.= +2.
B) C.N.= 5; O.N.= +3.
C) C.N.= 5; O.N.= +2.
D) C.N.= 6; O.N.= +3.
E) C.N.= 6; O.N.= +2.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 92
Related Exams